A standard blood pressure reading tells you something real. But it doesn’t tell you everything. Here is what your arm cuff misses about your brain, heart, and kidney health, and why it matters.
You checked your blood pressure. It came back normal. You moved on.
That reading told you something real. But it only measured pressure at your arm.

Medical Grade, Accurate, FDA-Cleared Heart Insights.
Save 20% on checkout with code VITALITY
Your heart, brain, and kidneys get their blood directly from or near the aorta, the main artery leaving the heart. Your arm is further down the arterial system. Pressure changes as blood travels, so those two readings are not always the same.
This article explains why they can diverge, what happens to those organs when they do, and what it means for how you think about cardiovascular monitoring.
What your arm cuff actually measures
When you wrap a cuff around your upper arm and take a reading, you are measuring brachial blood pressure. The brachial artery is the main artery running through your upper arm. The reading captures the force of your blood pushing against the wall of your blood vessel during each heartbeat.
This is a real and useful measurement. Brachial blood pressure has been the standard for decades, and it remains an important signal. But it is a peripheral measurement, taken at a point in the arterial system that is far from where your most vulnerable organs actually receive their blood.
Central blood pressure is different. It is the pressure in or near the aorta, where the heart first pushes blood out into the body. Your brain, heart muscle, and kidneys are all supplied by the aorta and its immediate branches. So central blood pressure reflects the force those organs actually experience with every beat.
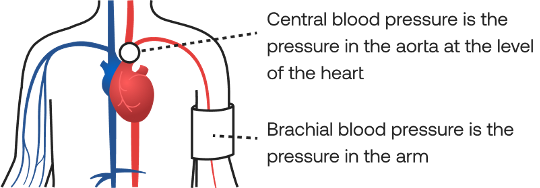
In a healthy arterial system, central and brachial pressures are related in a predictable way. The arm reading is a reasonable proxy for what is happening at the aorta.
When arteries stiffen, that relationship changes. Whether those two pressures still track together depends on what is happening in the arterial wall itself. That is what the rest of this article is about.
How your aorta is supposed to work
The aorta is not a rigid pipe. It is a flexible, elastic tube that does far more than simply carry blood.
With each heartbeat, the aorta stretches. It absorbs the surge of pressure as blood is pushed out of the heart, then recoils during the resting phase between beats. That cushioning action smooths the flow of blood as it travels through the body.
This matters for two reasons.
First, it reduces the workload on the heart. A flexible aorta gives way under pressure, which means the heart does not have to push against a rigid system with every contraction. Second, it prevents high-energy pressure waves from traveling too deep into the smaller, more delicate blood vessels in the brain and kidneys. Those vessels are not built to absorb the full force of each heartbeat. The aorta’s cushioning is what keeps that force from reaching them.
When the aorta stiffens, that cushion degrades. The heart has to work harder against a system that no longer gives way the same way. And more pulsatile energy, the force of each heartbeat, travels deeper into the microcirculation, the smallest blood vessels inside organs, that rely on the aorta to absorb it.
Several factors drive this stiffening over time. Aging is the most universal. Sustained high blood pressure accelerates it. So do diabetes, smoking, and long periods of physical inactivity. You can read more about what changes in the arterial wall as these factors accumulate. For this article, the focus is on what happens next.
Think of it as a shock absorber
Think of the aorta as the shock absorber between your heart and the rest of your arterial system. A new shock absorber is soft and pliable. It gives way smoothly under load, then recovers. An old, hardened shock absorber still works, but the force it was supposed to cushion now passes through to whatever is on the other side. Your aorta does the same job for every heartbeat, every day, for your entire life.
What happens to your brain
The brain receives roughly 20% of the body’s resting blood flow. It is supplied through a network of small, delicate vessels that carry that flow into and through the tissue itself. Those vessels are not built to absorb the full force of each heartbeat. The aorta is supposed to absorb it for them.
When the aorta can no longer do that job, the excess force reaches the brain’s smallest vessels. Over time, this is associated with measurable changes: lesions in the brain’s white matter, small vessel damage visible on imaging, and other signs of wear that appear long before symptoms do.
A 2023 meta-analysis of 37 studies covering nearly 28,000 people found a clear pattern. Higher arterial stiffness was associated with greater evidence of small vessel changes in the brain. The association with white matter changes specifically was even stronger.
These changes are linked to slower processing, memory changes, and a higher risk of vascular cognitive decline over the long term. They do not mean symptoms are imminent, and they do not predict any one person’s cognitive future. What they show is that arterial stiffness and brain health are more connected than a blood pressure reading alone would suggest.
The brain’s small vessels have no backup. When that protection is gone, they take the force directly. That is the connection this research keeps surfacing.
What happens to your kidneys
The kidneys filter your blood, and doing that job requires the highest blood flow rate of any major organ relative to their size. They also have the lowest vascular resistance, meaning blood passes through their network of small vessels with very little obstruction.
That combination is what makes them vulnerable. High-volume, low-resistance flow is efficient for filtration, but it also means the kidneys have very little built-in protection against excess pulsatile force. When that protection breaks down, the kidneys are among the first organs exposed to the consequences.
Research tracking patients over time has established that the relationship between arterial stiffness and kidney damage runs in one direction. Arterial stiffness contributes to kidney damage, not just the other way around. A 2024 study of over 10,000 adults applied a statistical method designed to separate cause from coincidence by tracking the same people over several years. They found that increased arterial stiffness actually precedes the decline in kidney function, and that individuals with elevated arterial stiffness had more than double the risk of developing chronic kidney disease.
That finding matters because it closes a loop that observational research alone cannot close. Earlier studies had shown the two conditions move together. This one suggests one is contributing to the other.
What that looks like in practice is a gradual decline in how well the kidneys filter waste from the blood. It is not dramatic. It accumulates over years. And the earliest signs of it rarely show up in a standard blood pressure reading.
What happens to your heart
The heart is on both sides of this problem.
It is the organ generating the pressure. It is also the organ most immediately affected when the system it pushes against stiffens.
A flexible aorta gives way with each contraction. The heart pushes, the aorta stretches, and the workload stays manageable. When the aorta stiffens, that flexibility disappears. The heart has to push harder against a system that no longer absorbs the force. Over time, that increased workload can cause the heart muscle to thicken and work less efficiently.
There is a second effect that is less obvious. The heart muscle itself receives most of its own blood supply during diastole, the brief resting phase between beats. That is when the coronary arteries fill. A stiff aorta reduces diastolic pressure, which means less blood reaches the heart muscle during the only window it has to receive it.
So the heart is asked to work harder while receiving less of the blood supply it needs to sustain that work. That imbalance does not produce sudden symptoms. It builds gradually. And it is one of the pathways through which arterial stiffness is linked to heart failure risk over the long term.
A standard blood pressure reading captures the pressure the heart is generating. It does not capture how hard the heart is working to generate it, or whether the heart is getting what it needs in return.
Why your arm reading doesn’t capture this
Everything in the previous three sections, the excess force reaching the brain, the pulsatile damage accumulating in the kidneys, the increased workload on the heart, is happening at or near the aorta and its branches. Not at the arm.
The pressure at your arm and the pressure at the aorta are related. But they are not identical. In a healthy, flexible arterial system, the difference between them is relatively consistent. A brachial reading is a reasonable proxy for what is happening centrally.
As arteries stiffen, that proxy becomes less reliable. Central blood pressure can rise while brachial blood pressure stays stable or changes less dramatically. The two readings can also diverge in the other direction. The arm reading looks the same. The pressure your organs experience does not.
A 2025 study of more than 34,000 adults found that roughly 1 in 27 participants had elevated central blood pressure while their standard brachial reading appeared normal. Their cardiovascular event risk was comparable to people whose readings were elevated on both measures. A standard reading alone could not tell them which group they were in.
This is not an edge case. In a population of that size, 3.65% represents a meaningful number of people whose risk was invisible to the measurement they relied on.
Central blood pressure is also more strongly associated with markers of organ damage than brachial blood pressure is. Left ventricular changes, arterial wall thickening, arterial stiffness itself: in each case, the evidence shows a stronger relationship with central pressure than with the reading taken at the arm. The same body of research confirms that central blood pressure and arterial stiffness predict cardiovascular risk and mortality more reliably than peripheral arm readings alone.
The arm cuff is not wrong. It is incomplete. And the gap between what it captures and what your organs experience is exactly what central blood pressure and arterial stiffness measurement is designed to reveal.
What more complete monitoring looks like
The practical question is straightforward: how do you see the rest?
Annual blood pressure measurement at the arm is a starting point. It is not the finish line.
Adding central blood pressure and arterial stiffness to what you track gives you visibility into the signals this article has been describing: the pressure your organs actually experience, whether the aorta is still absorbing the force of each heartbeat, and whether those signals are stable or shifting over time.
Measurement is one part of the picture. What moves these signals in the right direction is consistent cardiovascular care: regular physical activity, sleep, stress management, and attention to the factors that accelerate arterial stiffening. What the research shows about how quickly those habits translate into measurable improvement is more encouraging than most people expect.
Seeing what a standard reading misses
The CONNEQT Pulse tracks central blood pressure and arterial stiffness at home. The cuff captures the pressure waveform at your arm. Using pulse wave analysis, it derives what that pressure looks like closer to the heart and the organs it supplies.
The method has been validated against catheter-based measurements taken directly inside the aorta and used in clinical settings worldwide for more than two decades. Not a diagnostic tool. A way to see whether the signals that an arm cuff cannot capture are stable, improving, or moving in the wrong direction over time.
None of this is a reason to alarm yourself about a single reading. The associations in this article are long-term relationships between arterial function and organ health. They develop over years and decades, not days. What they are is a reason to widen the lens. Cardiovascular risk does not only live in the numbers a brachial cuff can see.
Earlier visibility into the signals that matter gives you time, context, and a more complete conversation with your clinician. That is the case for tracking more than one number.
What to read next
This article covered what happens to your brain, heart, and kidneys when arterial stiffness changes the pressure they experience, and why a standard blood pressure reading cannot capture that process. The articles below go deeper into the topics this piece touched on.
- Early Signs of Arterial Stiffness: What Changes Before Symptoms Appear – What changes in the arterial wall before symptoms appear, and what earlier visibility into those changes can do for you.
- Why Arterial Stiffness Doesn’t Wait for High Blood Pressure – Why arterial changes can develop before blood pressure rises, and what that means for how you monitor your cardiovascular health.
- When Is Blood Pressure Actually High? – How to think about blood pressure readings in context, and what patterns matter more than any single number.
- Three Ways to Look at Your Arteries, And Why You Need All of Them – A complementary look at arterial health through structure, flow, and function.
- How Pressure Amplification Influences Your Arterial Health Readings – Why the pressure at your arm and the pressure at your aorta can diverge, and what that divergence tells you about your arterial health.
Want to explore more? Browse the full library.